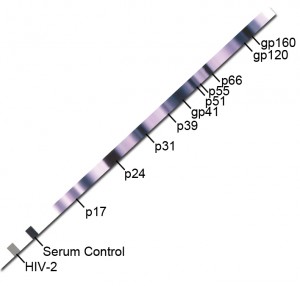
More specific tests such as viral isolation or molecular probes may be necessary to distinguish between infections with these viruses in certain populations. Because of the serological cross reactivities between HIV-1 and HIV-2, in certain populations it is difficult to ascertain whether an individual is infected with HIV-1, HIV-2, a new viral type, or whether the individual is infected simultaneously with multiple viruses. Nine of the 42 specimens were discordant by Western blot and RIPA classification, being dually reactive by one procedure and reactive with only one viral gp120 by the other technique. Among these 33 specimens, 10 were found to be reactive for antibodies to HIV-1 only, 10 were reactive to HIV-2 only, and 13 were considered to be dually reactive, having antibodies reactive with both HIV-1 gp120 and HIV-2 gp120. With confirmatory Western blot, the chance of a false-positive identification in a low-prevalence setting is about 1 in 250 000 (95 CI, 1 in 173 000 to 1 in 379 000). In specimens from West Africa and Portugal, Western blot and RIPA were in complete agreement on 33 of 42 samples (78.6%). The HIV proteins used in western blotting can be produced by recombinant DNA in a technique called recombinant immunoblot assay (RIBA). Antibody reactivity to the envelope protein gp120 was able to discriminate 20 of 20 (100%) U.S. Extensive serologic cross reactivity between HIV-1 and HIV-2 p24 was found in both populations. Western blots and sodium dodecylsulfate-polyacrylamide gel electrophoresis (SDS-PAGE) radioimmunoprecipitation assays (RIPA) utilizing native HIV-1 and HIV-2 proteins were performed on these specimens to determine the ability of these procedures to discriminate between HIV-1 and HIV-2 infections. The remainder require a maximum of 3 months' follow-up to resolve anti-HIV-1 status.Serum and plasma samples were collected from blood donors who were confirmed positive for antibodies to HIV-1 in the United States, and from blood donors and individuals in West Africa and Portugal who were positive for antibodies to HIV-1, HIV-2, or both. For in vitrodiagnostic use 32508 40 Tests FOR REFERENCE USE ONLY: DO NOT USE in place of package inserts provided with each test kit. It is consistent with the HIV diagnostic algorithm proposed by the Centers for Disease Control and Prevention 2. Grouping HIV-1 WB-indeterminates according to profile allows a significant percentage to be reported as anti-HIV-1-negative, while additional testing may allow others to be reported as anti-HIV-1-positive. A Western Blot Kit for the Detection of Antibodies to Human Immunodeficiency Virus Type 1 (HIV-1) in Human Serum, Plasma or Dried Blood Spots. HIV-1/2 Antigen and Antibodies, Fourth Generation, with Reflexes - This test is used to help diagnose HIV-1 and HIV-2 infection, including acute infection, and to differentiate HIV-1 from HIV-2. The other two groups required additional testing and/or follow-up to distinguish reactivity caused by anti-HIV-1 from cross-reactivity. Samples in two out of the four indeterminate groups were negative for anti-HIV-1. When testing failed to clarify anti-HIV-1 status, follow-up samples were taken to monitor changes in antibody status. Objective: To reduce the number of HIV-1 Western blot (WB)-indeterminates requiring follow-up and the time taken to provide a clear positive or negative result. In the second stage, implementation of this strategy was assessed.Īfter dividing indeterminates into four groups according to WB profile, samples were tested for anti-HIV-1, anti-HIV-2, anti-HTLV-I antibodies, and HIV-1 antigen using the most sensitive assays available. In the first of two stages, a testing and follow-up strategy was developed to resolve anti-HIV-1 status of WB-indeterminates. To reduce the number of HIV-1 Western blot (WB)-indeterminates requiring follow-up and the time taken to provide a clear positive or negative result.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
